Nitrocalcite: Difference between revisions
Jump to navigation
Jump to search
Weblinks
No edit summary |
|||
| Line 40: | Line 40: | ||
<br clear=all> | <br clear=all> | ||
{|border="2" cellspacing="0" cellpadding="4" width="52%" align="left" class="wikitable" | {|border="2" cellspacing="0" cellpadding="4" width="52%" align="left" class="wikitable" | ||
|+''Table 1: Deliquescence humidities of nitrocalcite at different round temperatures <bib id="Steiger etal: 2014"/>'' | |+''Table 1: Deliquescence humidities of nitrocalcite at different round temperatures <bib id="Steiger.etal:2014"/>'' | ||
|- | |- | ||
|bgcolor = "#F0F0F0" align=center| 0°C | |bgcolor = "#F0F0F0" align=center| 0°C | ||
| Line 57: | Line 57: | ||
|} | |} | ||
<br clear=all> | <br clear=all> | ||
==Under the polarizing microscope == | ==Under the polarizing microscope == | ||
Revision as of 17:05, 15 February 2015
| Nitrocalcite[1] | |

| |
| Mineralogical name | Nitrocalcite |
| Chemical name | Calcium Nitrate Tetrahydrate |
| Trivial name | Nitrate of lime |
| Chemical formula | Ca(NO3)2•4H2O |
| Other forms | |
| Crystal system | monoclinic |
| Crystal structure | |
| Deliquescence humidity 20°C | 53.1% [Steiger etal: 2014]Title: Weathering and Deterioration Author: Steiger, Michael; Charola A. Elena; Sterflinger, Katja 
|
| Solubility (g/l) at 20°C | 2660 g/l |
| Density (g/cm³) | 1.82 g/cm3 |
| Molar volume | 129.8 cm3/mol |
| Molar weight | 236.16 g/mol |
| Transparency | transparent |
| Cleavage | |
| Crystal habit | |
| Twinning | |
| Phase transition | |
| Chemical behavior | |
| Comments | |
| Crystal Optics | |
| Refractive Indices | nx = 1.465 ny = 1.498 nz = 1.504 |
| Birefringence | Δ = 0.039 |
| Optical Orientation | negative |
| Pleochroism | |
| Dispersion | |
| Used Literature | |
back to Nitrate
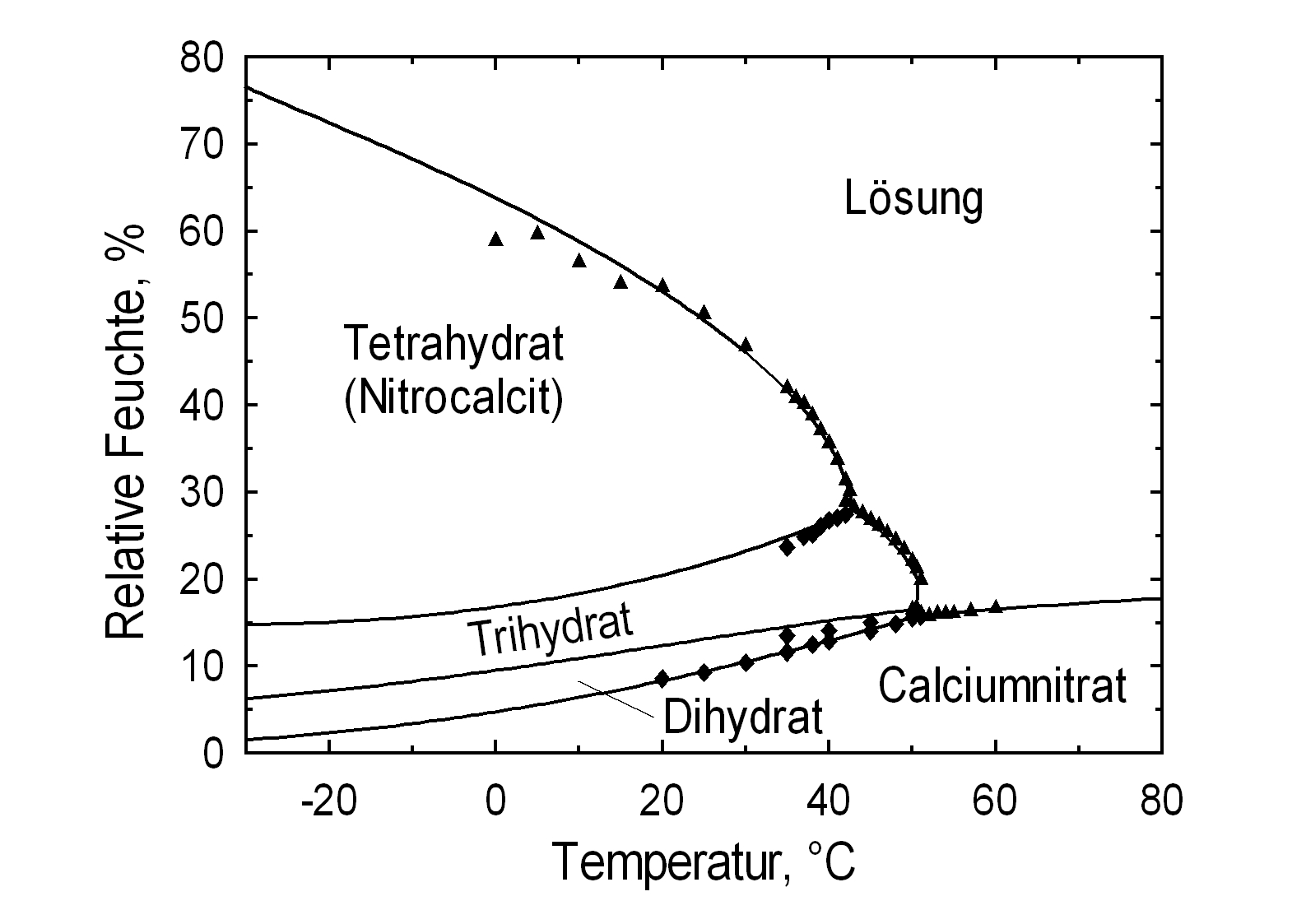
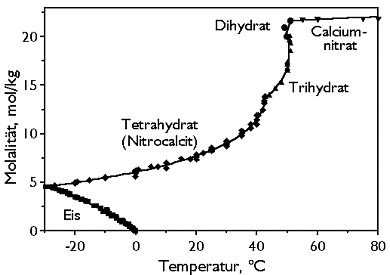
Phase Diagrams[edit]
Hygroscopicity[edit]
| 0°C | 10°C | 20°C | 30°C | 40°C | 50°C |
| 63.8%r.h. | 58.8%r.h. | 53.1%r.h. | 46.0%r.h. | 35.5%r.h. | 21.3%r.h. (Ca(NO3)2•3H2O) |
Under the polarizing microscope[edit]
- Crystallized from a saturated solution with ethanol addition
- Crystallized in a micro climate chamber
Weblinks
[edit]
- ↑ http://www.mindat.org/min-2919.html seen on 29.07.2010
Literature[edit]
| [Broul.etal:1981] | Elsevier (eds.) Broul M., Nyvlt J.; Soehnel O. (1981): Solubility in organic two component systems, Elsevier |  |
| [Steiger.etal:2014] | Steiger, Michael; Charola A. Elena; Sterflinger, Katja (2014): Weathering and Deterioration. In: Siegesmund S.; Snethlage R. (eds.): Stone in Architecture, Springer Verlag Berlin Heidelberg, 223-316, 10.1007/978-3-642-45155-3_4. |  |