Talk:Thenardite: Difference between revisions
SLeithaeuser (talk | contribs) |
SLeithaeuser (talk | contribs) No edit summary |
||
| Line 115: | Line 115: | ||
== Hydration pressure == | == Hydration pressure == | ||
The hydration pressure, which occurs at the transition from thenardite to [[mirabilite]], is highly dependent on the existing relative humidity and temperature conditions. This correlation is shown in table 3 as follows: | |||
<br clear="all"> | <br clear="all"> | ||
{|border="2" cellspacing="0" cellpadding="4" width="52%" align="left" class="wikitable" | {|border="2" cellspacing="0" cellpadding="4" width="52%" align="left" class="wikitable" | ||
|+'' | |+''Table 3''': Hydration pressure thenardite-[[mirabilite]] nach <bib id="Winkler.etal:1970"/>'' | ||
|- | |- | ||
|bgcolor = "#F0F0F0" align=center| ''' | |bgcolor = "#F0F0F0" align=center| '''RH %''' | ||
|bgcolor = "#F0F0F0" align=center| '''20.0 °C''' | |bgcolor = "#F0F0F0" align=center| '''20.0 °C''' | ||
|bgcolor = "#F0F0F0" align=center| '''25.0 °C''' | |bgcolor = "#F0F0F0" align=center| '''25.0 °C''' | ||
| Line 158: | Line 158: | ||
<br clear=all> | <br clear=all> | ||
The change in volume, taking place during phase transition, is to be specified at approx. 320%. | |||
<bib id="Sperling.etal:1980"/>. | |||
== Analytical detection == | == Analytical detection == | ||
| Line 164: | Line 165: | ||
=== Microscopy<br> === | === Microscopy<br> === | ||
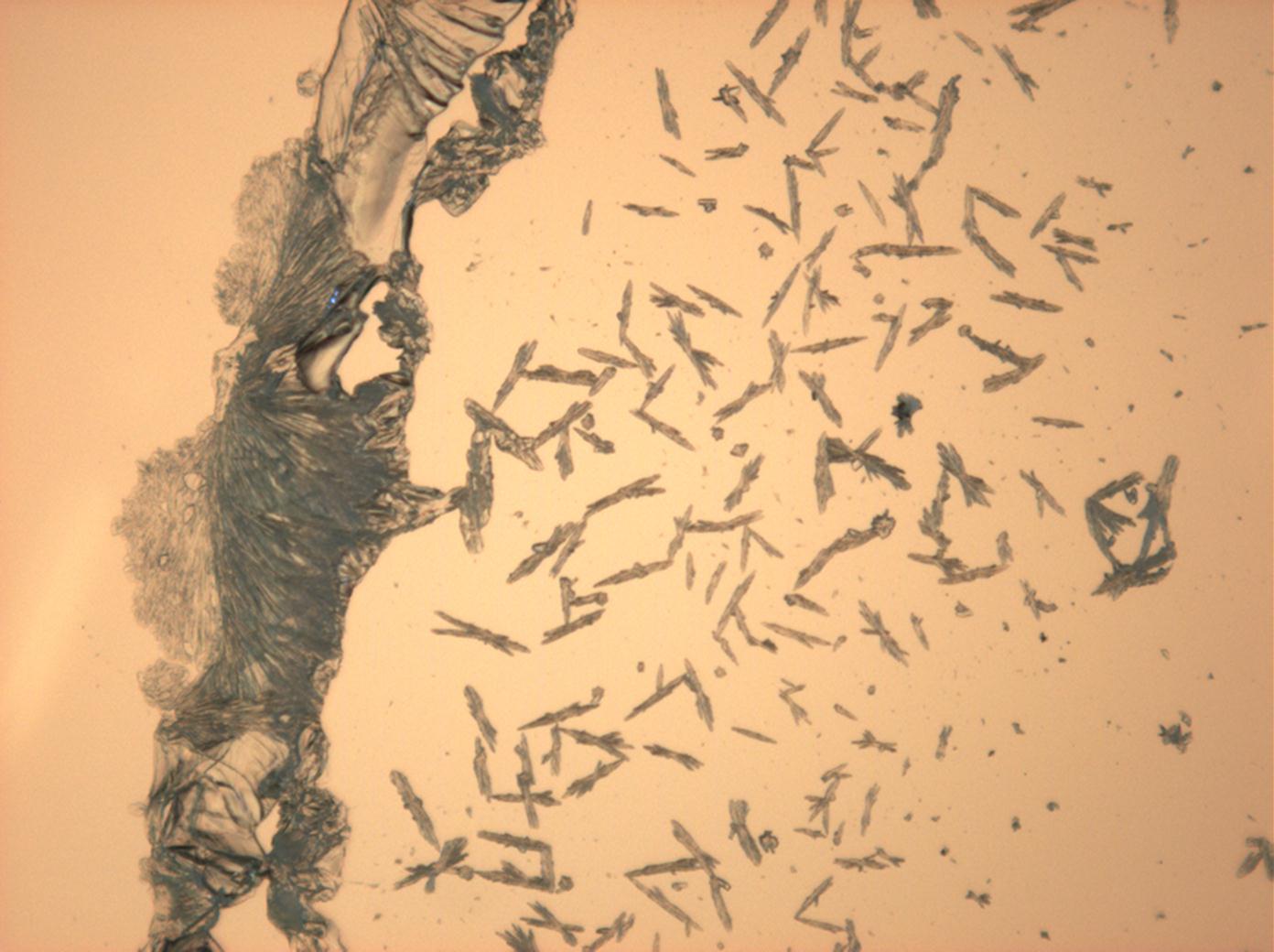
''' | '''Laboratory investigation:'''<br> Through microscopic observations regarding the solubility behavior, good solubility in water and no solubility in ethanol can be confirmed. Thenardite and mirabilite do not have morphological characteristics that could help identification with the use of simple re-crystallization experiments. | ||
<br> | |||
''' | '''Refractive indices:''' n<sub>x</sub> = 1.468; n<sub>y</sub> =1,473; n<sub>z</sub> =1.483<br>'''Birefringence''': Δ = 0.015<br>'''Crystal class''': orthorhombic<br> | ||
<br> | <br> | ||
'''[[ | '''[[Polarized light microscopy]] examination:'''<br> | ||
The raw sampling material and the re-crystallized preparation change their water of crystallization content, depending on the conditions of relative humidity and temperature. | |||
In dry air conditions (RH < 80% and room temperature) [[mirabilite]] looses its chemically bound water and changes to become thenardite. This process can be clearly understood and reproduced using a microscopic, when the process of re-crystallization is observed. Mirabillite does not show any characteristic abnormal interference colors. During the moisture loss and the formation of thenardite the abnormal interference phenomena become weaker.<br><br>The refractive index assignment of thenardite is carried out using the immersion method. Due to the low maximum birefringence, thenardite mostly displays gray interference colors. The extinction is parallel or symmetrical. | |||
<br>''' | <br>'''Possible mistakes:''' | ||
Generally, the differentiation between certain kinds of sulfates (they are listed below, thenardite included) is a problem without micro-chemical determination of the anions, because the refractive indices of the salts lie very closely together and all salts show a low birefringence. It is best to use an immersion medium with a n<sub>D/sub>- value of 1,48, thus making the differentiation within this group possible. Moreover, the properties mentioned below can be consulted as criteria for determination. | |||
Thenardite is unambiguously, but indirectly determined by re-crystallizing a sample and observing the abnormal interference colors, which occur when mirabiltite is identified in its high hydrate form. | |||
{|border="2" cellspacing="0" cellpadding="4" width="100%" align="left" class="wikitable" | {|border="2" cellspacing="0" cellpadding="4" width="100%" align="left" class="wikitable" | ||
|+'' | |+''Table 3''': Characteristics for differentiating thenardite from other sulfates'' | ||
|- | |- | ||
|bgcolor = "#F0F0F0"| ''' | |bgcolor = "#F0F0F0"| '''Salt phase''' | ||
|bgcolor = "#F0F0F0"| ''' | |bgcolor = "#F0F0F0"| '''Characteristic''' | ||
|- | |- | ||
|bgcolor = "#F7F7F7"| '''[[ | |bgcolor = "#F7F7F7"| '''[[Boussingaultite]]''' (NH<sub>4</sub>)<sub>2</sub>Mg(SO)<sub>4</sub> • 6H<sub>2</sub>0 | ||
|bgcolor = "#FFFFEO"| | |bgcolor = "#FFFFEO"| no abnormal interference colors/ inclined extinction | ||
|- | |- | ||
|bgcolor = "#F7F7F7"| '''[[Schönit|Pikromerit]]''' K<sub>2</sub>Mg(SO<sub>4</sub>)<sub>2</sub> • 6H<sub>2</sub>0 | |bgcolor = "#F7F7F7"| '''[[Schönit|Pikromerit]]''' K<sub>2</sub>Mg(SO<sub>4</sub>)<sub>2</sub> • 6H<sub>2</sub>0 | ||
|bgcolor = "#FFFFEO"| | |bgcolor = "#FFFFEO"| no abnormal interference colors/ inclined extinction | ||
|- | |- | ||
|bgcolor = "#F7F7F7"|'''[[Astrakanit|Bloedit]]''' Na<sub>2</sub>Mg(SO<sub>4</sub>)<sub>2</sub> • 6H<sub>2</sub>0 | |bgcolor = "#F7F7F7"|'''[[Astrakanit|Bloedit]]''' Na<sub>2</sub>Mg(SO<sub>4</sub>)<sub>2</sub> • 6H<sub>2</sub>0 | ||
|bgcolor = "#FFFFEO"| | |bgcolor = "#FFFFEO"| all indices >1.48 / no abnormal interference colors/ inclined extinction / optically negative orientation. | ||
|- | |- | ||
|bgcolor = "#F7F7F7"| '''[[Aphthitalit|Glaserit]]''' K<sub>3</sub>Na(SO<sub>4</sub>)<sub>2</sub> | |bgcolor = "#F7F7F7"| '''[[Aphthitalit|Glaserit]]''' K<sub>3</sub>Na(SO<sub>4</sub>)<sub>2</sub> | ||
|bgcolor = "#FFFFEO"| | |bgcolor = "#FFFFEO"| all indizes >1.48 / no abnormal interference colors/ inclined extinction | ||
|- | |- | ||
|bgcolor = "#F7F7F7"| '''[[ | |bgcolor = "#F7F7F7"| '''[[Arcanite]]''' K<sub>2</sub>SO<sub>4</sub> | ||
|bgcolor = "#FFFFEO"| | |bgcolor = "#FFFFEO"| all indices >1.48 / no abnormal interference colors | ||
|- | |- | ||
|bgcolor = "#F7F7F7"| '''[[ | |bgcolor = "#F7F7F7"| '''[[Magnesium formiate]]''' Mg(HCO<sub>2</sub>)<sub>2</sub> • 2H<sub>2</sub>O | ||
|bgcolor = "#FFFFEO"| | |bgcolor = "#FFFFEO"| comparatively high birefringence / no abnormal interference colors/ inclined extinction | ||
|} | |} | ||
<br> | <br> | ||
''' | '''Observation of mixed systems:''' | ||
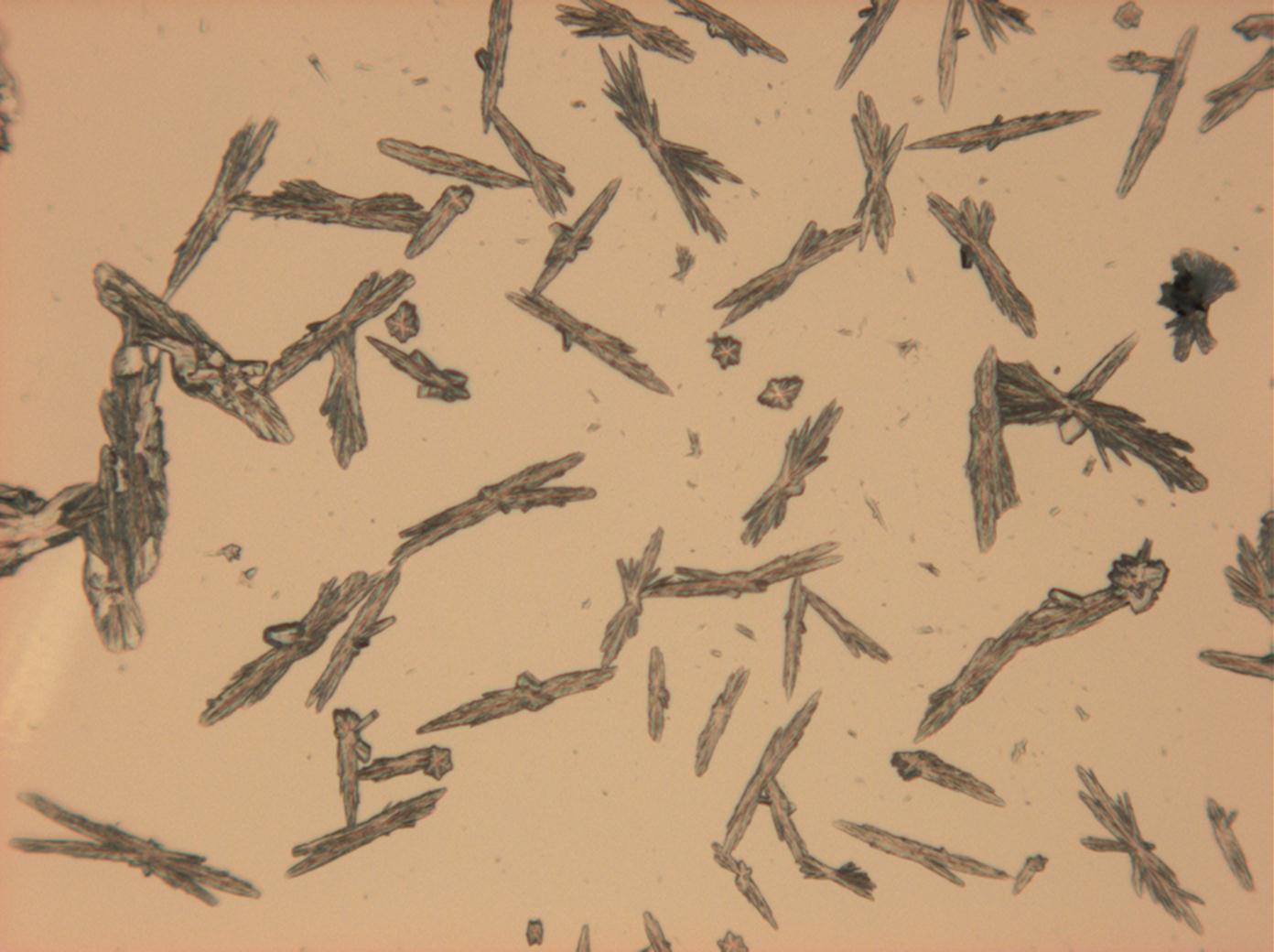
The mixed system Na<sup>+</sup>– Ca<sup>2+</sup>– SO<sub>4</sub> <sup>2-</sup>: The precipitation of [[gypsum]] takes place first during re-crystallization, which is due to its low solubility. The distinct needle like habit of single gypsum crystals and aggregates remains. | |||
The precipitation of sodium sulfate takes place later. The actual crystal growth takes place much faster. The morphology is non-specific. | |||
Mixed system Na<sup>+</sup>– SO<sub>4</sub> <sup>2-</sup>– Cl<sup>-</sup>: The precipitation of both kinds of particle starts approximately at the same time, [[halite]] with its characteristic morphology, [[sodium sulfate]] in extremely varying forms. | |||
Revision as of 10:09, 1 June 2012
| Thenardite[1][2] | |

| |
| Mineralogical name | Thenardite |
| Chemical name | Sodium sulfate |
| Trivial name | Pyrotechnite |
| Chemical formula | Na2SO4 |
| Other forms | Mirabilite (Na2SO4•10H2O) Sodiumsulfate heptahydrate (Na2SO4•7H2O) |
| Crystal system | orthorhombic |
| Crystal structure | |
| Deliquescence humidity 20°C | 81.7% (25°C) |
| Solubility (g/l) at 20°C | 162 g/l |
| Density (g/cm³) | 2.689 g/cm³ |
| Molar volume | 53.11 cm3/mol |
| Molar weight | 142.04 g/mol |
| Transparency | transparent to translucent |
| Cleavage | perfect |
| Crystal habit | |
| Twinning | |
| Phase transition | |
| Chemical behavior | |
| Comments | soluble in water and glycerin, not soluble in pure alcohol |
| Crystal Optics | |
| Refractive Indices | nx = 1.468 ny = 1.473 nz = 1.483 |
| Birefringence | Δ = 0.015 |
| Optical Orientation | positive |
| Pleochroism | |
| Dispersion | |
| Used Literature | |
| {{{Literature}}} | |
Authors: Hans-Jürgen Schwarz , Michael Steiger, Tim Müller
English translation by Matthieu Angeli
back to Sulfate
Sodium sulfate and thenardite[edit]
Abstract[edit]
Sodium sulfate and its phases thenardite, mirabilite and heptahydrate are described. Heptahydrate´s role as a major source of damage is also explained .
Occurrence[edit]
Both thenardite and mirabilite occur as natural minerals. In nature sodium sulfate occurs in mineral waters in the form of double salts, as deposits of former salt lakes. Knowledge of the hydrated sodium sulfate dates back to the 16th Century. Its first description has been written by Glauber in 1658 in which he described it as "sal mirable". It is also quite common to read the name "Glauber's salt" for mirabilite in the literature.
Information on the origin and formation of thenardite / mirabilite in monuments[edit]
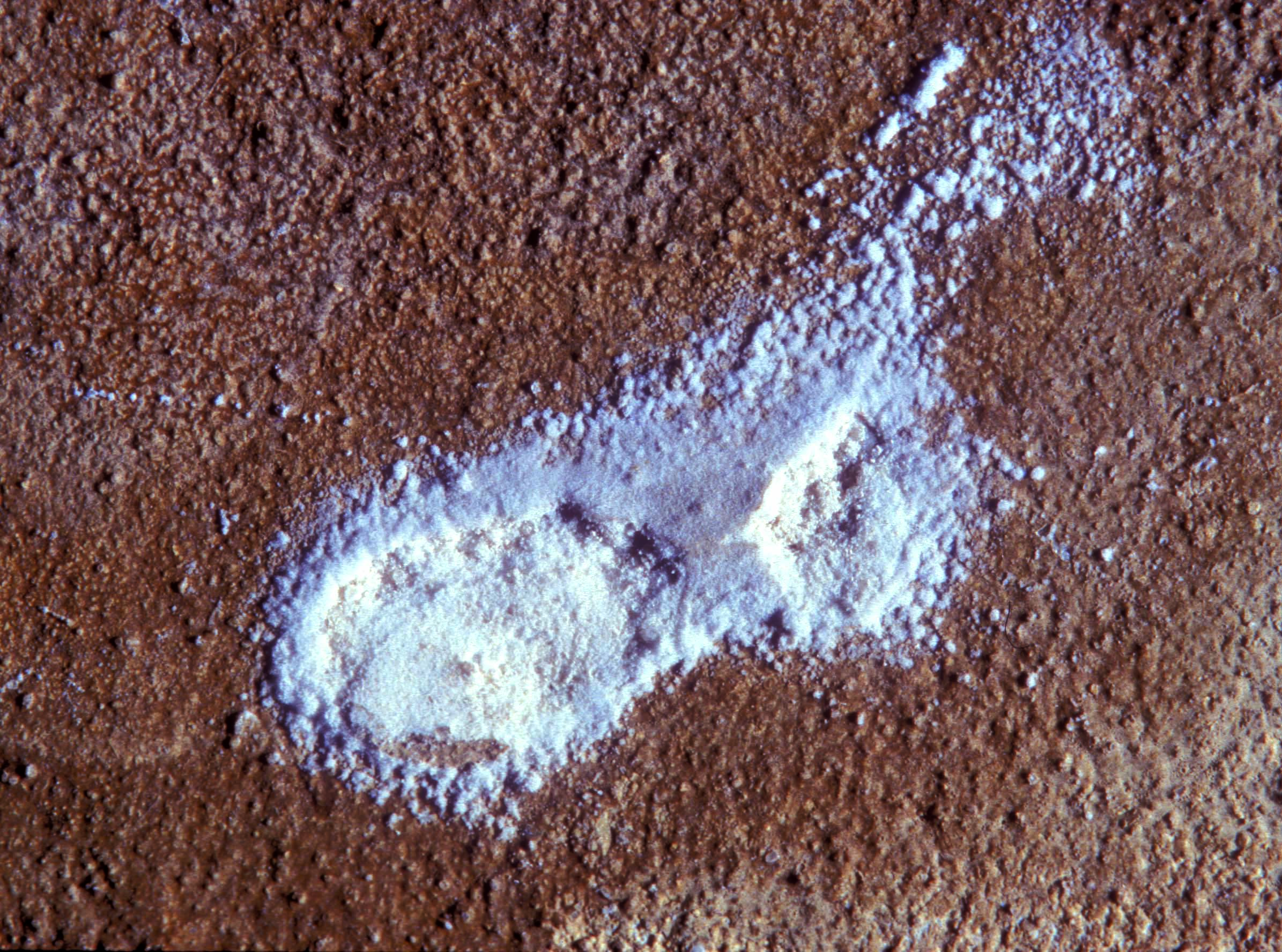
With the entry of materials that contain soluble sodium compounds, the mineral system of a monument may create sodium sulfate as salt efflorescence, when reacting with various sources of sulfate for example air contaminated with sulphurous gases. In Germany cement exhibits a high content of sodium ions, because the German standardization institute (DIN) allows for a content of up to 0.5 % soluble alkalis. This means that 100 kg of Portland cement containing only 0.1% soluble Na2O can form 520g of Mirabilite when in contact with air containing sulfuric acid [calculation by Arnold/Zehnder 1991]. Sodium ions can also enter into monuments from a plethora of cleaning materials and especially older restoration products (such as water glass). Ground water and surface water are also a possible source of Na+-ions. Road salt consists to a large extent of slightly soluble sodium chloride. Finally, in coastal areas, sea water is a significant source of NaCl.
Solubility behavior[edit]
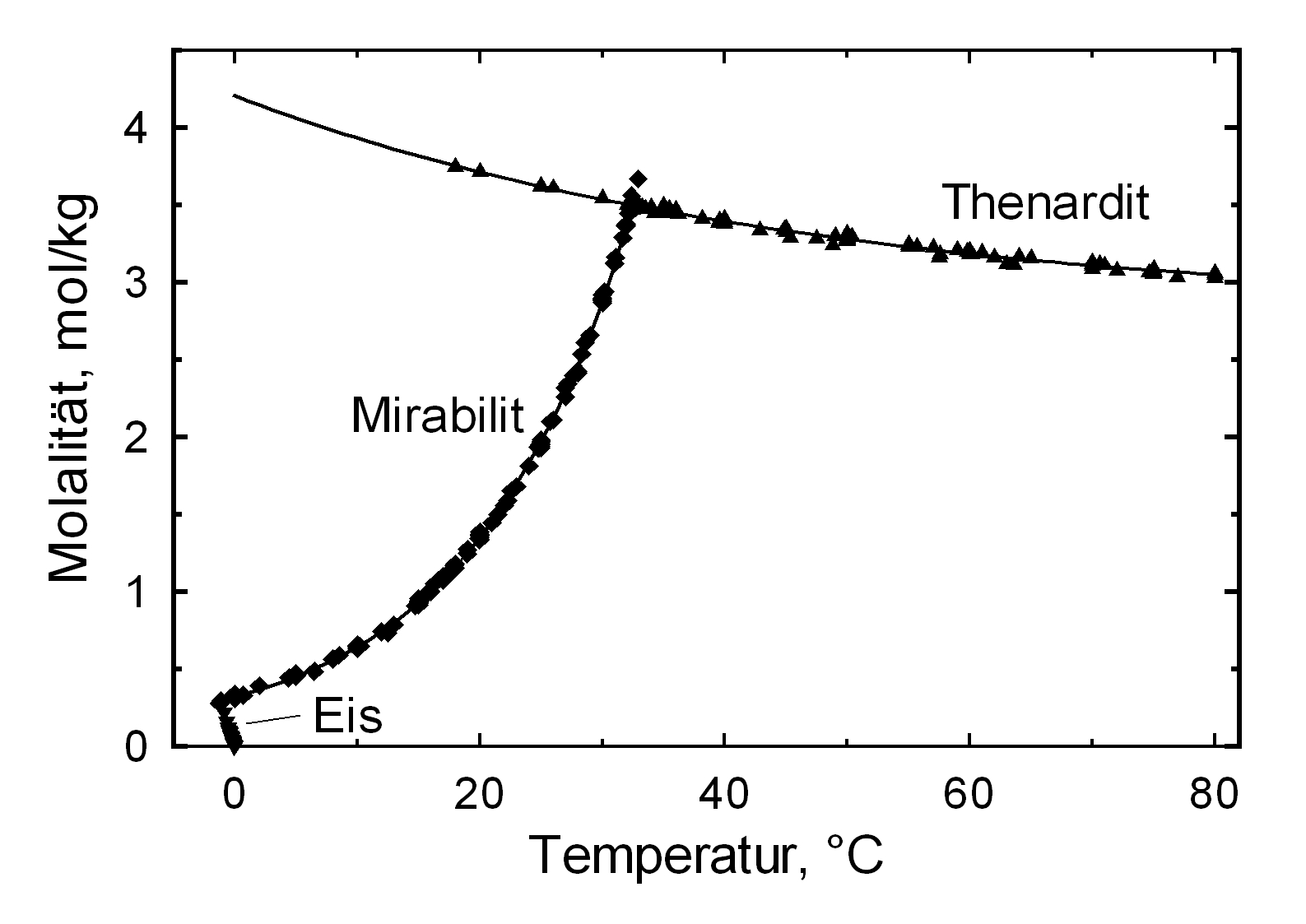
The structures of both thenardite and mirabilite belong to the group of easily soluble salts and therefore they are easily mobilized (see table hygroscopicity of the salts and ERH). The solubility of sodium sulfate is highly dependent on temperature. For this reason, a rapid drop of temperature is highly likely to yield very high supersaturation and salt crystallization.
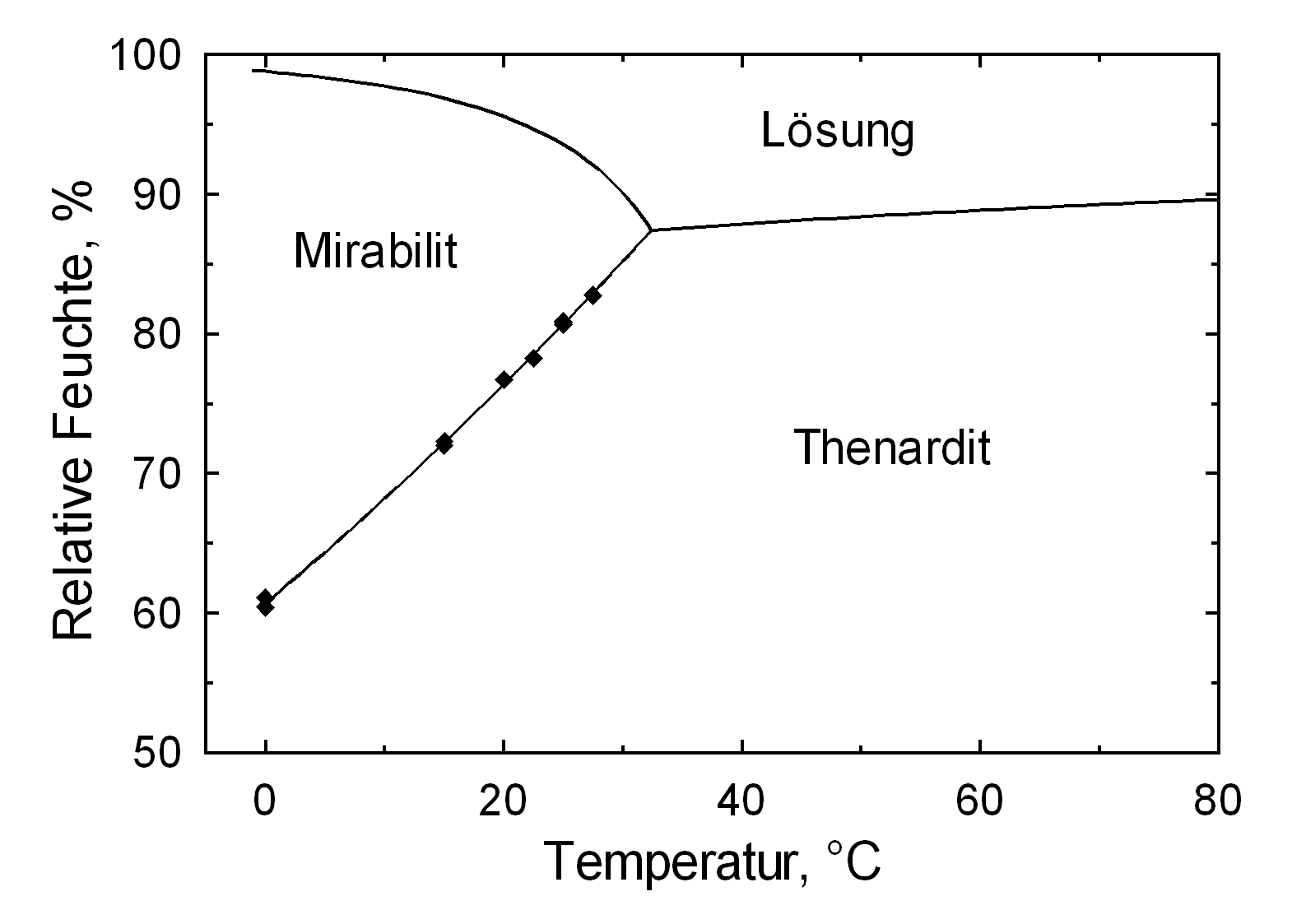
Hygroscopicity[edit]
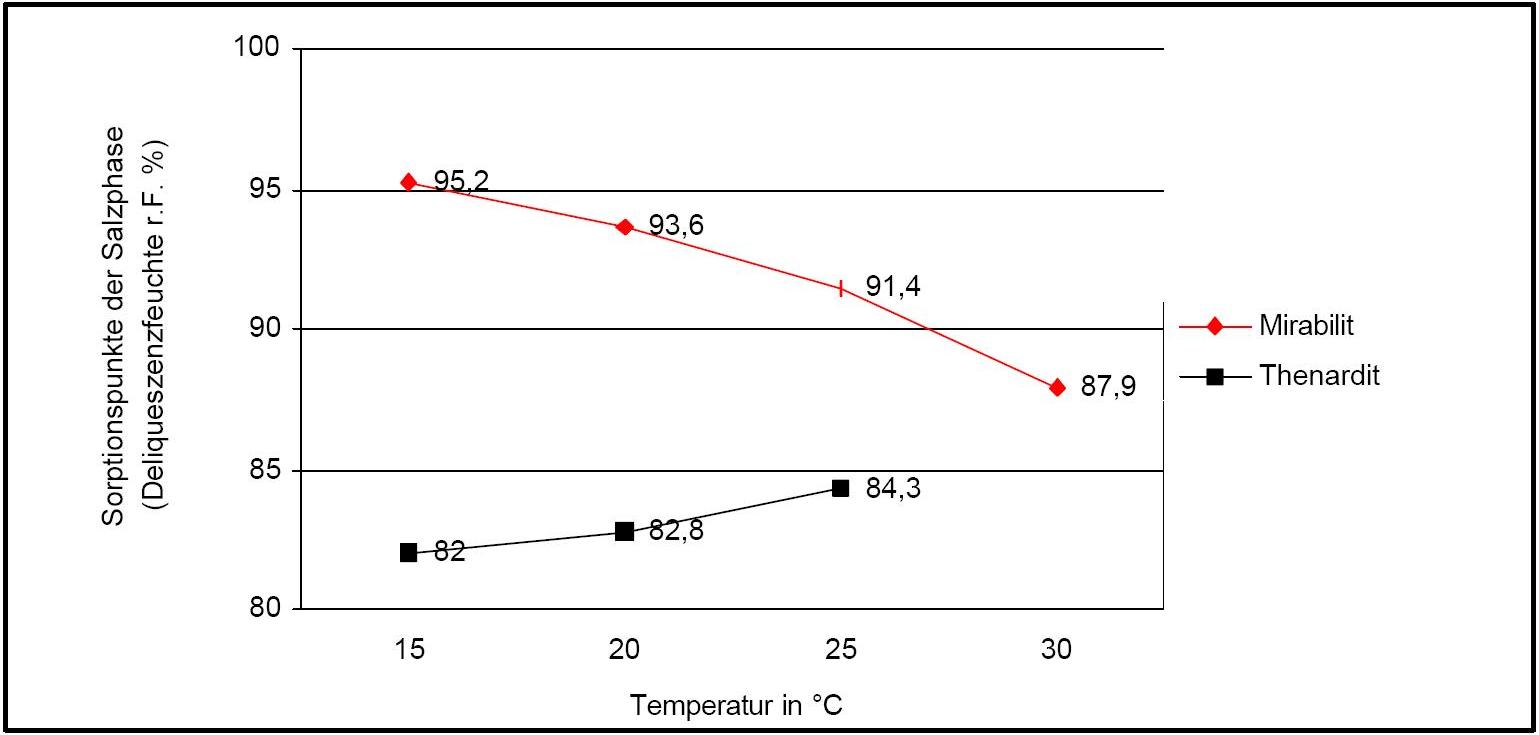
The temperature effect on the deliquescence points of thenardite and mirabilite is shown below. The striking features here are the opposite curve transitions. In the presence of other ions (in salt mixtures), the parameters of the equilibrium moisture content, as well as the necessary temperature and humidity conditions for recrystallization, change significantly. The following table shows experimental data of equilibrium moisture for different salt mixtures at different temperatures.It turns out that all the values of equilibrium moisture content are lower than those of the pure salt mirabilite (see table equilibrium moisture content as a function of temperature).
| MgSO4 | Ca(NO3)2 | KNO3 | |
| Na2SO4 • 10H2O | 87(21°C) | 74 (21°C) | 81(21°C) |
Water vapor sorption:
Author: Arnold, Andreas; Zehnder, Konrad

The table below shows additional information for estimating the hygroscopicity of sodium sulfate for the sorption behavior of pure salt and the mixture with halite at different relative humidity levels:
| Air humidity | 87% r.F. | 81% r.F. | 79% r.F. |
| Na2SO4 | 79 | 0 | 0 |
| Na2SO4+NaCl (1:1 molar mixture) | 157 | 32 | 15 |
Crystallization pressure[edit]
When crystallizing from an aqueous solution, the crystallization pressure of thenardite lies in the 29.2-34.5 N/mm2 range. These values are higher that those calculated for other building-damaging salts [Winkler:1975]Title: Stone: Properties, Durability in Man´s Environment
Author: Winkler, Erhard M. .
.
Hydration behavior[edit]
The Na2SO4 – H2O system:
The only stable forms of sodium sulfate are the decahydrate (mirabilite) and the anhydrite (thenardite). The generation of mirabilite by recrystallization of the salt from an aqueous supersaturated solution occurs at 32.4°C. In particular, the transition from thenardite to mirabilite and the incorporation of 10 water molecules in the crystal lattice causes a volume expansion of 320%. This transition happens at a relatively low temperature (32-35°C), the damage caused by this salt is highly dependent on the temperature and thus on the environment. This temperature range is given as a guide, because this transition could happen for example at 25°C at 80% relative humidity, or even at 0°C at 60.7% relative humidity [information from Gmelin]. Due to this strong dependence on the environmental parameters, an estimate of the damage caused on buildings by crystallization and hydration of sodium sulfate are very difficult to obtain.
Pictures of salt and salt damage[edit]
In the field[edit]
- Thenardite efflorescences
Under the polarizing microscope[edit]
- Sodium sulfate crystals between to glass plates
- Sodium sulfate crystals, crystallised out of a water extraction of a real sample
Weblinks
[edit]
- ↑ http://webmineral.com/data/Thenardite.shtml viewed on 29/07/2010
- ↑ http://www.mindat.org/min-3935.html viewed on 29/07/2010
Literature[edit]
There were no citations found in the article. SLeithaeuser 15:22, 6 May 2012 (CEST)